A) 0.045 L
B) 0.28 L
C) 0.56 L
D) 0.90 L
E) 1.1 L
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In a redox reaction,the reducing agent undergoes loss of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2H2(g) + O2(g) 2H2O(l)
B) Zn(s) + H2SO4(aq) ZnSO4(aq) + H2(g)
C) H2O(l) + NH3(g) NH4+(aq) + OH-(aq)
D) 6FeSO4(aq) + K2Cr2O7(aq) + 7H2SO4(aq)
Cr2(SO4) 3(aq) + 3Fe2(SO4) 3(aq) + K2SO4(aq) + 7H2O(l)
E) Cl2(g) + 2KBr(aq) Br2(l) + 2KCl(aq)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction.
2NaCl(l) 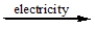 2Na(l) + Cl2(g)
2Na(l) + Cl2(g)
A) acid-base
B) precipitation
C) combination
D) displacement
E) decomposition
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
In both of the following reactions,a precipitate is formed.Complete and balance the equations,showing the phases of the products. a.AgNO3(aq)+ CaCl2(aq) b.NaOH(aq)+ Fe(NO3)3(aq)
Correct Answer

verified
a.2AgNO3(aq)+ CaCl<...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The oxidation numbers of P,S and Cl in H2PO2-,H2S and KClO4 are,respectively
A) -1,-1,+3
B) +1,-2,+7
C) +1,+2,+7
D) -1,-2,+7
E) -1,-2,+3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
In each of the following cases,write down the oxidation number of the indicated atom. a.P in P4 b.C in C2H6 c.S in H2SO4 d.Mn in MnO4- e.S in S4O62- f.P in Na3PO4
Correct Answer

verified
a.0
b.-3
c...View Answer
Show Answer
Correct Answer
verified
b.-3
c...
View Answer
Multiple Choice
Predict the products by completing a balanced equation for the following decomposition reaction.
CaCl2(l) 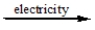 ?
?
A) CaCl2(l) ![]()
Ca(l) + 2Cl-(l)
B) CaCl2(l) ![]()
Ca2+(l) + Cl2(g)
C) CaCl2(l) ![]()
Ca2+(l) + 2Cl-(l)
D) CaCl2(l) ![]()
Ca(l) + Cl2(g)
E) CaCl2(l) ![]()
CaCl(l) + Cl-(l)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the net ionic equation for the reaction between lithium hydroxide and hydrobromic acid. LiOH(aq) + HBr(aq) H2O(l) + LiBr(aq)
A) LiOH(aq) Li+(aq) + OH-(aq)
B) HBr(aq) H+(aq) + Br-(aq)
C) H+(aq) + OH-(aq) H2O(l)
D) Li+(aq) + Br-(aq) LiBr(aq)
E) Li+(aq) + OH-(aq) + H+(aq) + Br-(aq) H2O(l) + LiBr(aq)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the oxidation number of sulfur in sodium metabisulfite,Na2S2O5.
A) -2
B) +2
C) +4
D) +5
E) None of these is the correct oxidation number.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is a redox reaction?
A) 2Na(g) + Cl2(g) 2NaCl(s)
B) Ba2+(aq) + SO42-(aq) BaSO4(s)
C) K2Cr2O7(aq) + 2KOH(aq) 2K2CrO4(aq) + H2O(l)
D) Na2CO3(s) + 2HCl(aq) 2NaCl(aq) + CO2(g) + H2O(l)
E) H2O(l) H+(aq) + OH-(aq)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g)
B) H2(g) + Cl2(g) 2HCl(g)
C) 2H2O2(aq) 2H2O(l) + O2(g)
D) Fe2O3(s) + 3H2SO4(aq) Fe2(SO4) 3(aq) + 3H2O(l)
E) 2KMnO4(aq) + 10FeSO4(aq) + 8H2SO4(aq)
K2SO4(aq) + 2MnSO4(aq) + 5Fe2(SO4) 3(aq) + 8H2O(l)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Vinegar is a solution of acetic acid,CH3COOH,dissolved in water.A 5.54-g sample of vinegar was neutralized by 30.10 mL of 0.100 M NaOH.What is the percent by weight of acetic acid in the vinegar?
A) 0.184%
B) 1.63%
C) 3.26%
D) 5.43%
E) 9.23%
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
a.Explain or define what is meant by the term "oxidation".
b.Write down the oxidation numbers of the atoms in the following formulas.

Correct Answer

verified
a.Oxidation: loss of...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
In a redox reaction,the oxidizing agent undergoes loss of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a weak acid?
A) H2SO4
B) HNO3
C) HF
D) HBr
E) HCl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. KOH(aq) + HCl(aq) KCl(aq) + H2O(l)
A) precipitation
B) acid-base
C) redox
D) combination
E) None of these choices is correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2H2O2(aq) 2H2O(l) + O2(g)
A) combination
B) decomposition
C) displacement
D) acid-base
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction.
CaCl2·H2O(s) 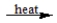 CaCl2(s) + H2O(g)
CaCl2(s) + H2O(g)
A) combination
B) decomposition
C) displacement
D) acid-base
E) precipitation
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
In both of the following reactions,a precipitate is formed.Complete and balance the equations,showing the phases of the products. a.BaCl2(aq)+ Na2SO4(aq) b.Mg(NO3)2(aq)+ KOH(aq)
Correct Answer

verified
a.BaCl2(aq)+ NaView Answer
Show Answer
Correct Answer
verified
View Answer
Showing 61 - 80 of 84
Related Exams