A) s
B) p
C) d
D) f
E) g
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization state of the carbon (I) atom in the following compound? 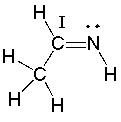
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to molecular orbital theory the highest energy molecular orbital that is occupied with an electron is referred to as _____.
A) degenerate
B) antibonding
C) the LCAO
D) the LUMO
E) the HOMO
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate bond angle around the indicated carbon atom? 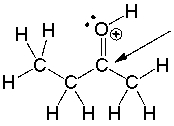
A) 600
B) 900
C) 109.50
D) 1200
E) 1800
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization state of the nitrogen atom in the following compound? 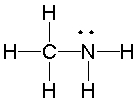
A) sp
B) sp2
C) sp3
D) sp3d
E) s2p
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the formal charges on boron and oxygen in the following structure? 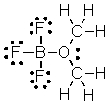
A) B = 1-, O = 1-
B) B = 1-, O = 1+
C) B = 1+, O = 1+
D) B = 1+, O = 1-
E) B = 1-, O = 0
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization state of the indicated atoms in the following compound? 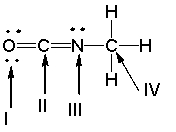
A) I - sp ; II - sp2 ; III - sp3 ; IV - sp2
B) I - sp2 ; II - sp ; III - sp2 ; IV - sp3
C) I - sp3 ; II - sp2 ; III - sp ; IV - sp2
D) I - sp2 ; II - sp3 ; III - sp2 ; IV - sp
E) I - sp2 ; II - sp2 ; III - sp2 ; IV - sp3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures have a 1- formal charge on the sulfur atom? 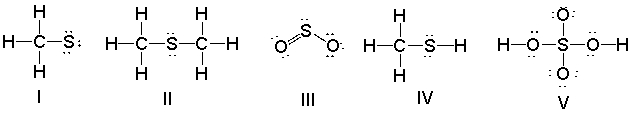
A) I
B) II
C) III
D) IV
E) V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which intermolecular force is primarily responsible for base pairing, and stability, of the double helix in DNA?
A) ion-dipole interactions
B) London dispersion forces
C) dipole-dipole interactions
D) hydrogen bonding
E) covalent bonding
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following ground state electron configuration violates ____. 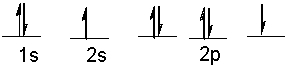
A) the Aufbau principle
B) the Pauli Exclusion principle
C) Hund's Rule
D) Heisenberg's Uncertainty principle
E) None of these.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Interaction of the following two atomic orbitals results in what kind of molecular orbital, in the orientation shown? 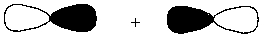
Correct Answer

verified
sigma bond...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following covalent bonds has the largest dipole moment?
A) C-C
B) C-H
C) C-O
D) N-H
E) H-F
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds have the lowest boiling point? 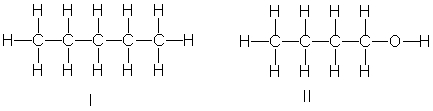
A) I
B) II
C) III
D) IV
E) II and IV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
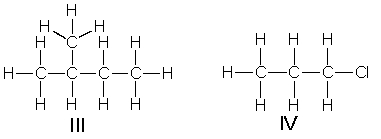
Which of the following structures have carbon with sp2 hybridization state? 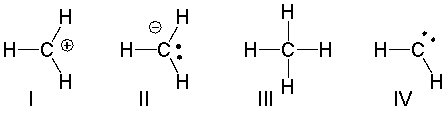
A) I and II
B) III and IV
C) I and III
D) II and IV
E) I and IV
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds have trigonal planar molecular geometry? 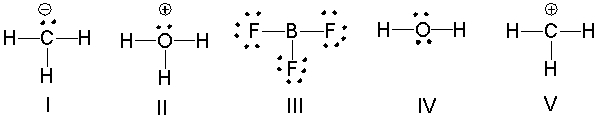
A) I, II and III
B) II and III
C) III and V
D) V only
E) All of these
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
For NaSCH3, identify each bond as polar covalent, nonpolar covalent or ionic.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electronegativity of elements on the periodic table tends to increase_______.
A) from left to right, top to bottom
B) from right to left, bottom to top
C) from left to right, bottom to top
D) from right to left, top to bottom
E) from upper right to lower left
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statement is incorrect, when the 1s atomic orbitals of two hydrogen atoms results in constructive interference?
A) a sigma bonding molecular orbital is formed
B) the bonding molecular orbital formed is lower in energy than the 1s atomic orbital
C) the bonding molecular orbital formed has a node between the atoms
D) the bonding molecular orbital formed has circular symmetry
E) a maximum of two electrons may occupy the bonding molecular orbital
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sigma bonds are present in the following compound? 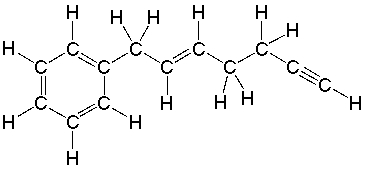
A) 20
B) 22
C) 24
D) 25
E) 27
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct Lewis structure for SCN- including the formal charges, if any? 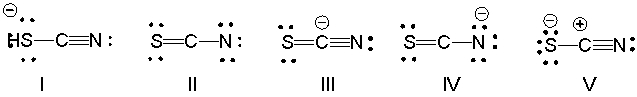
A) I
B) II
C) III
D) IV
E) V
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 191
Related Exams