Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum forms a layer of aluminum oxide when exposed to air which protects the bulk metal from further corrosion. 4Al(s) + 3O2(g) 2Al2O3(s) Calculate G° for this reaction, given that G°f of aluminum oxide is -1576.4 kJ/mol.
A) -3152.8 kJ/mol
B) -1576.4 kJ/mol
C) -788.2 kJ/mol
D) 1576.4 kJ/mol
E) 3152.8 kJ/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the temperature at which Kp = 4.00 for the reaction N2O4(g) ![Find the temperature at which K<sub>p</sub> = 4.00 for the reaction N<sub>2</sub>O<sub>4</sub>(g) 2NO<sub>2</sub>(g) .[Given: at 25°C, for NO<sub>2</sub>(g) , \Delta H°<sub>f</sub> = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N<sub>2</sub>O<sub>4</sub>(g) , \Delta H°<sub>f</sub> = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that \Delta H° and \Delta S° are independent of temperature.] A) 197 °C B) 56 °C C) 36 °C D) 79 °C E) 476°C](https://d2lvgg3v3hfg70.cloudfront.net/TB3245/11ea7cc5_01bc_7214_a2ab_a1a03afd5e2c_TB3245_11.jpg) 2NO2(g) .[Given: at 25°C, for NO2(g) , H°f = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N2O4(g) , H°f = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that H° and S° are independent of temperature.]
2NO2(g) .[Given: at 25°C, for NO2(g) , H°f = 33.85 kJ/mol, S° = 240.46 J/mol·K; for N2O4(g) , H°f = 9.66 kJ/mol, S° = 304.3 J/mol·K; assume that H° and S° are independent of temperature.]
A) 197 °C
B) 56 °C
C) 36 °C
D) 79 °C
E) 476°C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The heat of vaporization of water is 2.27 kJ/g. What is Svap per mole at the normal boiling point?
Correct Answer

verified
Correct Answer
verified
Short Answer
Hvap for ethanol is 38.7 kJ/mol at its boiling point (78°C).What is Ssurr when 1.00 mole of ethanol is boiled?
Correct Answer

verified
Correct Answer
verified
Short Answer
For the reaction CuS(s)+ H2(g)  H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= - 20.6 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= - 20.6 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?
Correct Answer

verified
No
Correct Answer
verified
Multiple Choice
For the reaction HCONH2(g)  NH3(g) + CO(g) , Kc = 4.84 at 400 K. If H° for this reaction is 29 kJ/mol, find Kc at 500 K.
NH3(g) + CO(g) , Kc = 4.84 at 400 K. If H° for this reaction is 29 kJ/mol, find Kc at 500 K.
A) 5.8
B) 0.17
C) 27
D) 0.88
E) 10.3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For a given substance the entropy always increases in the following order: S (gas)< S (liq)< S (solid).
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of solid naphthalene is introduced into an evacuated flask. Use the data below to calculate the equilibrium vapor pressure of naphthalene (C10H8) in the flask at 35°C. 
A) 890.mmHg
B) 0.21 mmHg
C) 696 mmHg
D) 0.086 mmHg
E) 833 mmHg
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange these reactions according to increasing S. 1) H2O(g) H2O(l) 2) 2NO(g) N2(g) + O2(g) 3) MgCO3(s) MgO(s) + CO2(g)
A) 1 < 2 < 3
B) 2 < 3 < 1
C) 3 < 2 < 1
D) 2 < 1 < 3
E) 1 < 3 < 2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
For the reaction SbCl5(g)  SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?
SbCl3(g)+ Cl2(g),
G°f (SbCl5)= -334.34 kJ/mol
G°f (SbCl3)= -301.25 kJ/mol
H°f (SbCl5)= -394.34 kJ/mol
H°f (SbCl3)= -313.80 kJ/mol
Will this reaction proceed spontaneously at 298 K and 1 atm pressure?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the normal boiling point of triethylborane (C6H15B) using the following data: 
A) 92°C
B) -21°C
C) 21°C
D) 365°C
E) 256°C
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate Kp at 298 K for the reaction SO2(g) + NO2(g) SO3(g) + NO(g) . 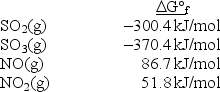
A) 6.99 10-7
B) 5.71 10-8
C) 14.2
D) 475
E) 1.42 106
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate S° for the reaction SO2(s) + NO2(g) SO3(g) + NO(g) . 
A) 53.6 J/K·mol
B) -53.6 J/K·mol
C) -22.2 J/K·mol
D) 474.8 J/K·mol
E) -474.8 J/K·mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Using the thermodynamic data provided below, calculate Ka2 for H2CO3(aq)at 25°. 
Correct Answer

verified
4.59  10-11
10-11View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Rubidium has a heat of vaporization of 69.0 kJ/mol at its boiling point (686°C).Calculate S for this process, Rb(l) Rb(g), at 1 atm and 686°C.
Correct Answer

verified
Correct Answer
verified
True/False
The entropy change S° for the reaction NH4Cl(s) NH3(g)+ HCl(g)will be negative.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The entropy change on vaporization ( Svap) of a compound or element is
A) always negative.
B) always positive.
C) sometimes is positive and sometimes is negative.
E) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
The element oxygen was prepared by Joseph Priestley in 1774 by heating mercury(II) oxide: HgO(s) \9\to\) Hg(l) + 1/2O2(g) , H° = 90.84 kJ/mol. Estimate the temperature at which this reaction will become spontaneous under standard state conditions. S°(Hg) = 76.02 J/K·mol S°(O2) = 205.0 J/K·mol S°(HgO) = 70.29 J/K·mol
A) 108 K
B) 430 K
C) 620 K
D) 775 K
E) 840 K
G) C) and D)
Correct Answer

verified
E
Correct Answer
verified
Essay
Choose the substance with the higher entropy per mole at a given temperature: 1 mole of N2(g)in a 22.4 L container or 1 mole of N2(g)in a 2.24 L container.
Correct Answer

verified
1 mole of ...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 1 - 20 of 112
Related Exams