Correct Answer

verified
No. In addition to increasing other forms of strain, a planar conformation would make this molecule antiaromatic.
Correct Answer
verified
Short Answer
When cyclohexene is treated with KMnO4, H2O, the syn-1,2-diol is produced. What reaction occurs when benzene is similarly treated?
Correct Answer

verified
No reactio...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following compounds may correctly be classified as being aromatic? (More than one answer is possible.)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Provide an acceptable name for the compound below. 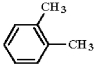
Correct Answer

verified
ortho-xyle...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Is the molecule below aromatic, antiaromatic, or nonaromatic? 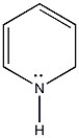
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures, if flat, would be classified as antiaromatic?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
An unknown compound gives the following spectral data. Propose a structure that is consistent with this information. In addition, show the fragmentations that give the 91 and 122 peaks in the MS. MS (m/z): 91, 108 and 122 (M+) 1H NMR (δ): 7.05(2H, d), 6.78 (2H, d), 3.7 (3H, s), 2.26 (3H, s)
Correct Answer

verified
Correct Answer
verified
Essay
Provide the structure of 4-isopropylbenzaldehyde.
Correct Answer

verified
Correct Answer
verified
Short Answer
Classify cyclopropenyl cation as aromatic, antiaromatic, or nonaromatic. Assume planarity of the π network
Correct Answer

verified
Correct Answer
verified
Short Answer
Provide an acceptable name for the compound below. 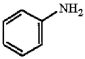
Correct Answer

verified
aniline or...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Describe the occupied π molecular orbitals in the ground state of cyclobutadiene.
Correct Answer

verified
Two electrons are paired in a ...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Provide the name of the compound shown below. 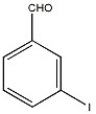
Correct Answer

verified
m-iodobenzaldehyde or 3-iodobenzaldehyde
Correct Answer
verified
Essay
Provide the structure of o-bromostyrene.
Correct Answer

verified
Correct Answer
verified
Essay
Provide the structure of sodium cyclopentadienide.
Correct Answer

verified
Correct Answer
verified
Essay
When toluene is treated with bromine (shown below), the bromine doesn't react with the double bond to form a viscinal dihalide, but instead a substitution of one of the benzylic hydrogens takes place. Why? 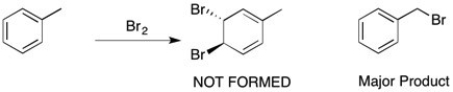
Correct Answer

verified
Addition of Br2 to the double bonds of be...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Show how the participating p orbitals interact to form the highest energy π molecular orbital of benzene.
Correct Answer

verified
All adjacent interactions are antibonding. 11eab5e1_c97c_31cc_9bae_cf288511523b_TB6199_00
Correct Answer
verified
Short Answer
Classify the compound below as aromatic antiaromatic, or nonaromatic. Assume planarity of the π network. 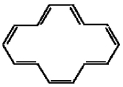
Correct Answer

verified
Correct Answer
verified
Essay
Circle and name the aromatic heterocycles in the following structure, which has been found to be effective against inflammatory diseases including Alzheimer's disease and rheumatoid arthritis (J. Med. Chem. 2007, 4728). 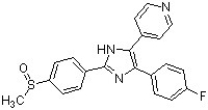
Correct Answer

verified
Correct Answer
verified
Short Answer
Is the molecule below aromatic, antiaromatic, or nonaromatic? 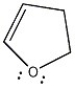
Correct Answer

verified
Correct Answer
verified
Essay
Is the all-cis form of [10]annulene aromatic? Explain.
Correct Answer

verified
No. Planarity, which...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 1 - 20 of 128
Related Exams